Wyoming Water Resources Center

|
RESEARCH BRIEFS Wyoming Water Resources Center |

|
| Technical RB92-01 |
Investigators: Keith T. Carron, Ken I. Mullen, Laura Peitersen, L. Gayle Hurley, and DaoXin Wang, Department of Chemistry, University of Wyoming.
Purpose: Current methods for groundwater analysis usually involve laboratory tests that require sample collection, transportation, and sophisticated laboratory equipment. This is time consuming, expensive, and often inaccurate due to volatilization of organics or oxidation of ionic species. An in-situ (on-site) method is being developed to accurately measure metals, pH and aromatic organic compounds in groundwater.
 Methods:
Methods for in-situ water quality analysis must have two characteristics,
selectivity for accurate contaminant identification and high sensitivity to
measure very low concentrations. The analytical technique applied in this
research to identify and measure groundwater contamination is called Raman
spectroscopy. The remote capabilities of our sensors come from the coupling
of Raman spectroscopy with optical fibers. Raman spectroscopy is a
vibrational technique that uses the frequency of scattered light to identify
molecules. Individual chemical compounds can be identified through
characteristic bands of frequencies that are specific to a particular
molecular structure. Raman is ideal for use with optical fibers since,
unlike infrared spectroscopy, it uses visible radiation that can be more
readily transmitted through fibers. High sensitivity is achieved through a
variation of Raman spectroscopy known as Surface Enhanced Raman Spectroscopy
(SERS). A million fold enhancement of Raman light scattering can be achieved
with specially roughened silver surfaces. Furthermore, silver coated SERS
probes overcome a major problem of our approach to remote sensing with
optical fibers by filtering out unwanted laser light prior to its entrance
into the collection fiber. These sensors can be placed several hundred
meters (e.g. down a well) from measurement instruments providing a new method
for in-situ analysis of groundwater.
Methods:
Methods for in-situ water quality analysis must have two characteristics,
selectivity for accurate contaminant identification and high sensitivity to
measure very low concentrations. The analytical technique applied in this
research to identify and measure groundwater contamination is called Raman
spectroscopy. The remote capabilities of our sensors come from the coupling
of Raman spectroscopy with optical fibers. Raman spectroscopy is a
vibrational technique that uses the frequency of scattered light to identify
molecules. Individual chemical compounds can be identified through
characteristic bands of frequencies that are specific to a particular
molecular structure. Raman is ideal for use with optical fibers since,
unlike infrared spectroscopy, it uses visible radiation that can be more
readily transmitted through fibers. High sensitivity is achieved through a
variation of Raman spectroscopy known as Surface Enhanced Raman Spectroscopy
(SERS). A million fold enhancement of Raman light scattering can be achieved
with specially roughened silver surfaces. Furthermore, silver coated SERS
probes overcome a major problem of our approach to remote sensing with
optical fibers by filtering out unwanted laser light prior to its entrance
into the collection fiber. These sensors can be placed several hundred
meters (e.g. down a well) from measurement instruments providing a new method
for in-situ analysis of groundwater.
Results:
Metal ions, pH and aromatic organics have been successfully detected and
concentrations determined in laboratory tests using SERS. Metal ions were
detected by analyzing the Raman spectrum of metal ion indicators bound to
the silver substrate on probes. Most metal ion indicators contain groups
that combine with metal ions, thus changing the Raman spectrum of the
 indicator. One advantage of metal ion indicators is that they often contain
a carboxylic acid or sulfonic group which increases the water solubility of
the indicator. Laboratory test results indicate detection limits for copper
(Cu2+) to be in the low parts per billion range and lead (Pb2+) has been
detected at the parts per trillion level.
indicator. One advantage of metal ion indicators is that they often contain
a carboxylic acid or sulfonic group which increases the water solubility of
the indicator. Laboratory test results indicate detection limits for copper
(Cu2+) to be in the low parts per billion range and lead (Pb2+) has been
detected at the parts per trillion level.
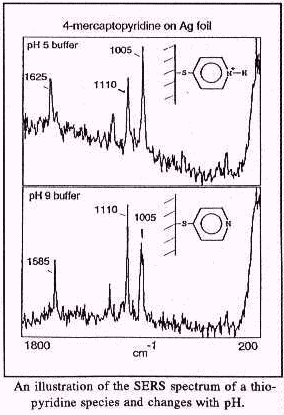
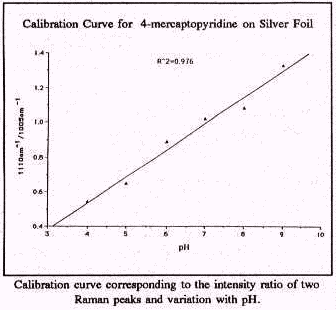
Probes to monitor pH were developed for two reasons. The first is to monitor the pH of groundwater. SERS sensors for pH produced excellent test results. A calibration curve was analyzed for variations in pH and relative Raman intensity for peaks associated with a protonated and with a deprotonated form of the pH indicator. Regression analysis of the calibration curve indicates a very close fit of the test data (a correlation coefficient or r2 of 0.98) using a probe with 4-mercaptopyridine on silver foil. The second reason for looking at pH at surfaces is to develop ways to buffer the surface pH such that large variations in the groundwater pH will not affect the probes. Furthermore, variation in surface pH is an excellent method for controlling the selectivity of the metal ion indicators. It should be possible to use a single metal ion indicator coupled with pH modified surfaces to fine tune the selectivity toward specific metal ions.
Future Research: Aromatic organic probes are being fabricated using the principle of partitioning. SERS substrates are modified with long chain alkylthiol coatings. The alkylthiols bond to the silver surface and create a hydrophobic coating. Organic compound which have a high lipid solubility (PCBs, DDT, PAH, etc.) partition into the surface coating and can then be detected using Raman spectroscopy. The detection limit will be determined by the partition coefficient of the organic substance. It is anticipated that part per trillion sensitivity can be obtained for compounds such as DDT and PCBs due to their very large partition coefficients. Future efforts are being directed at conducting field tests, commercial application and using SERS remote sensors to detect contaminants in vapor or gaseous phases.
Publications:
Carron, K., K. Mullen, M. Lanouette, and H. Angersbach. "Selective-Ultratrace Detection of Metal Ions with SERS," Applied Spectroscopy, 45, 420, (1991).
Mullen K. and K. Carron. "Surface Enhanced Raman Spectroscopy with Abrasively Roughened Fiber Optic Probes," Analytical Chemistry, 63, 2196, (1991).
Mullen, K., D. Wang, L. Hurley, and K. Carron. "Determination of pH: SERS Fiber Optic Probes." Forthcoming, Analytical Chemistry.
Carron, K. and L. Peitersen. "Detection of Organic Contaminants with Modified SERS Substrates." Forthcoming.
| For further information on this or other research projects or for a list of WWRC publications, telephone or write: |
Wyoming Water Resources Center P.O. Box 3067, University of Wyoming Laramie, WY 82071-3067 (307) 766-2143; FAX (307) 766-3718 |
WWRC RESEARCH BRIEFS are published by the Wyoming Water Resources Center with funds provided in part by the US Geological Survey, Department of Interior, as authorized by the Water Resources Research Act of 1984. The research on which this report is based was financed in part by the US Geological Survey, Department of Interior and Wyoming Water Resources Center. The views expressed do not necessarily represent those of the Department of Interior or the WWRC. Persons seeking admission, employment, or access to programs at the University of Wyoming shall be considered without regard to race, color, national origin, sex, age, religion, political belief, handicap, or veteran status.
 |
Technical RB92-01 |
Research Briefs List
Water Resources Data System Library |
Water Resources Data System Homepage